The Future Treatment for Parkinson’s Disease
Parkinson’s Disease
Parkinson’s disease is a progressive neurological disorder that affects 10 million people around the world and that number grows by 60,000 every year in the United States alone. Parkinson’s disease affects crucial functions in your brain responsible for body movement. Think of all the movements in your body, your hands, lips, legs, or back. Parkinson’s can affect all of these, leading to tremors, paralysis, rigidity and many other disabilities that Parkinson’s patients deal with every day.
The huge impact that Parkinson’s has on patients' lives, causes patients with Parkinson’s disease to suffer every day to do basic things like picking up a cup, trying to walk, hugging their children and many other simple tasks. The only way to treat Parkinson’s disease is to use medication or getting surgery. But both of the options have their downsides. Parkinson’s has no known cure, but we do know that Parkinson’s disease is caused by the lack of dopamine.
Economical Downside
Parkinson’s doesn’t just affect people suffering from Parkinson’s. But within the US, the combined direct and indirect cost of Parkinson’s, including treatment, social security payments and lost income is estimated to be nearly $52 billion per year. Medications alone cost an average of $2,500 a year per patient. Since medication is the primary treatment for all patients with Parkinson’s disease, the cost is about $25,000,000,000 spent on medication by all of the patients with Parkinson’s disease. The cost of medication and treatments alone can cause some patients to go into debt and create such a huge impact on their finances that some people go bankrupt.
Cause of Parkinson’s
The main cause of Parkinson’s is the lack of dopamine in the brain. The lack of dopamine is caused when nerve cells in the brain, which are mainly responsible for producing and relaying the neurotransmitter dopamine get damaged. So what is dopamine? Dopamine is a neurotransmitter that plays a key role in communication within your brain and other bodily functions, hence the name neurotransmitter. Dopamine transfers information from one part of the brain to another so a lack of dopamine creates an interruption between the transfer of information resulting in the signals not being carried to where they’re supposed to go.
The brain is like a circuit, everything has to work for it to function smoothly and effectively. The dopamine is similar to the wires of a circuit and when the wires can’t transfer electricity through the circuit, the circuit doesn’t work or is very ineffective. The same thing happens to dopamine, when there is a complete lack of dopamine the information is not transferred resulting in the body not work. In patients with Parkinson’s disease, their brains still produce dopamine but not at the level needed to function and this causes the brain to work ineffectively and results in the information not being transferred properly.
Current Solutions
The current solutions for Parkinson’s are mainly medication that patients have to take every day to artificially increase dopamine and surgeries in more serious cases which still require medication.
The most common medication is a pill called Levodopa that an average patient takes 3–6 times a day. Levodopa works by being ingested by the patient then it gets absorbed in the intestines and sent to the brain and once it reaches the brain barrier it converts into dopamine. Unfortunately, this has strongly been correlated with the rise to dyskinesia, which is an involuntary, jittery movement due to severe variation of dopamine levels in the brain. Dyskinesia is caused due to the severe variation in dopamine so when a patient has taken medication and forgets to take it afterwards or simply when the medication wears off, it creates a severe variation in dopamine levels.
Current surgical solutions, like Duopa and DBS, require your brain and body to be either very responsive to current medications or you must continue to go through surgical procedures for the coming years such as for battery replacements, and medication is still required. Duopa works by having a small incision in the stomach and has a tube placed in the intestine. The tube then pumps DUOPA which is a gel form of levodopa, allowing the dopamine to get to the brain faster by bringing it straight to the intestine. But DUOPA poses the same downsides as a medication since the medication is still involved it will still lead to dyskinesia. The DUOPA medication also costs a lot of money about an annual drug acquisition cost of approximately $61,000.
Deep brain stimulation (DBS) is an invasive surgery that places a neurostimulator in the brain that stimulates parts of the brain. DBS is not that well-known and we do not know how it works. There have been several proposed theories but none that prove how DBS works. DBS reduces the movement-related symptoms of Parkinson’s disease but patients are still required to take medication since the device isn’t creating dopamine in the brain.
Our Solution

But what if you could have a solution that doesn’t require you to regularly take medication and you don’t have to go through multiple surgical procedures in your lifetime?
The implant would be a surgical procedure where 2 sensors that measure dopamine and glutamate levels are placed in the nucleus accumbens of the brain, with wires attached to them that lead directly to the computer system securely placed outside the shoulder of the patient. There will also be a neurostimulator with its generator placed in the left shoulder and the leads going up the neck into the medial prefrontal cortex. The neurostimulator’s generator will also be connected to the computer placed on the shoulder. All components connected to the computer will be connected through wires which allow us to transfer data with little to no latency. The wire will be fed through a port to the outside of the body to protect the patient from developing infections and preventing anything to enter the body.
When abnormally low levels of dopamine are detected in the nucleus accumbens of the brain by our sensors, the installed neurostimulator will send out electrical impulses through attached leads at appropriate frequencies. The frequency will be determined by our software that will gradually increase the frequency until the sensors have picked up an increase, this will be known as the syncing period. As a result of the electrical impulses, there will be an increase in the overall levels of dopamine in the brain which will counteract the lack of dopamine that is hindering the basic functions of the brain. Using a computer will allow us to monitor and regulate the dopamine levels by increasing the dopamine at such a precise level automatically that there would be little to no variation preventing dyskinesia.
Our Product

We’ll be using a newly developed sensor at MIT that provides unmatched precision for measuring dopamine levels providing amazing potential for Parkinson’s treatment. By using microns only 10 mm in diameter and combined into arrays of eight electrodes, we can monitor dopamine levels at millisecond timescales. With older technologies, there is only about a 50 percent chance that a single electrode will end up in a spot where we can measure dopamine and that can also create scar tissues. This sensor will allow us to see the influence of the stimulation of the medial prefrontal cortex on the levels of dopamine and allows us to stimulate the dopamine at such precision that we can prevent dyskinesia.
We will also be using a sensor that measures glutamate levels in the nucleus accumbens of the brain. The sensor works by using a new sensor that detects ions in a solution, based on electric current or changes in electric current, which measures the random releases of glutamate in the brain. The glutamate biosensor will be used to measure the glutamate levels to make sure that our product does not increase the glutamate levels too much which could lead to seizures and the death of brain cells. If there was a case when glutamate levels rise to a very dangerous amount we could give the patients Lamotrigine, which is a release inhibitor of glutamate that is FDA-approved. If there was a case where high levels of glutamate existed the patient app will notify the patient.
The main aspect of our product consists of a neurostimulator that is currently being used to treat chronic pain in the spinal cord. The neurostimulator works by having the pacemaker-like device’s generator placed in the shoulder and then having the leads put up the neck into the medial prefrontal cortex of the brain. The leads will then send out electrical impulses at a certain frequency. The frequency will be determined automatically by the computer by having the computer increase the frequency gradually until a change in dopamine has been sensed by the dopamine sensor. The electrical impulses will stimulate the release of dopamine, glutamate and cholecystokinin. We are aiming to only increase dopamine so the stimulator will automatically stop at the designated level which will prevent too much dopamine in the brain. But if glutamate increases and gets to a dangerous level we can give our patient Lamotrigine only if the sensor detects a dangerous amount which our app will notify the patient to take the medication and see their doctor. The release of too much cholecystokinin leads to no known ill effects so our patients will not need to worry about the increase.
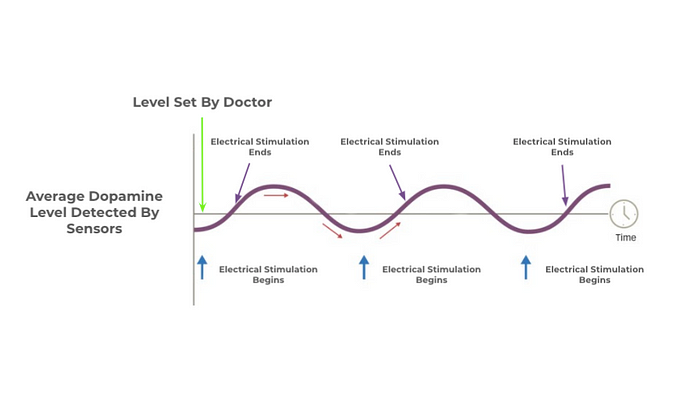
The device will also have a computer with programmable features on the outside of the body fixed to the shoulder on top of the generator which will connect to our device through a wire into the shoulder where there will be a port that will connect the inside components to the outside computer. The computer will monitor the dopamine levels 24/7 and once it drops below the designated level set by the doctor the computer will send out information to the neurostimulator. The neurostimulator will then send electrical impulses to the patient’s brain set to the frequency set by the syncing program. The impulses will increase dopamine levels reducing the effects of Parkinson’s disease. Once the dopamine has increased this process will be repeated every time the dopamine levels drop below the designated levels, automating that process by using the computer will reduce the variation of dopamine preventing dyskinesia. This will allow our patients to stop taking medication since we are increasing dopamine directly in the brain.
The device itself will be charged and powered through external batteries, allowing the patient to have this surgery once in their lifetime, if needed the battery can be replaced externally. The patient would have multiple batteries so while one battery is in use, the other ones would be charging and could alternate if notified by our application. Multiple batteries to power the device would allow the patient to travel long distances without worrying about the battery of the device.
With this precise dopamine monitoring system combined with a programmed neurostimulator that regulates dopamine levels in real-time, patients don’t have to worry about medication as well as the possibility of developing dyskinesia.
Application

The information collected as well as progress will be relayed to a patient through a patient application for full transparency. The patients will also be able to understand and see their progress. The application will also allow the patient to see the current battery level of the device and the patient will also be notified with low battery level alerts to let them know that they should change their batteries.
Commercial
Our product will be distributed and commercialized similar to how pacemakers and other tech in the healthcare industry are being incentivized right now. The process to get our product into the market would be through distributors in healthcare technology such as McKesson, Cardinal Health, and Medline Industries. Selling our product to hospitals through distributors will allow us to leverage existing partnerships that distributors have with hospitals already. Allowing us to get our product into the bodies of our consumers even faster.
We estimate to be able to build our product for approximately $21,000, which includes the neurostimulator, the leads, and the sensors. The reason for the high cost is due to the neurostimulator itself which costs about $17,000 alone. We then estimate that the computer itself will cost about $1,500. The sensors we hope to use are not available for customer use so we are estimating that they cost $3,000. We believe that our product will have the ability to monitor and regulate dopamine at such a precision that isn’t able to be done by current solutions for a fraction of the cost.
The Future
With constant development and improvement of technologies and solution systems, Parkinson’s patients will be able to live life easier than ever before. Our solution will allow our patients to forget that they even had Parkinson’s in the first place by allowing them to forget about medication and dyskinesia. In the future, we would like the implant and device to be used for several other mental illnesses such as depression, anxiety, and ADHD. Our product will completely change the treatments for mental health conditions and make patients suffering from mental health conditions feel better about themselves and improve their quality of life.
Please feel free to contact us at the following email: sharu.sivapalan@gmail.com
